Teva Branded Pharmaceutical Products R&D;, . An electronic inhaler add-on module which store and transmit information related to inhaler use (eModule) PDR0000401 FCC ID 2AJVSPDR0000401
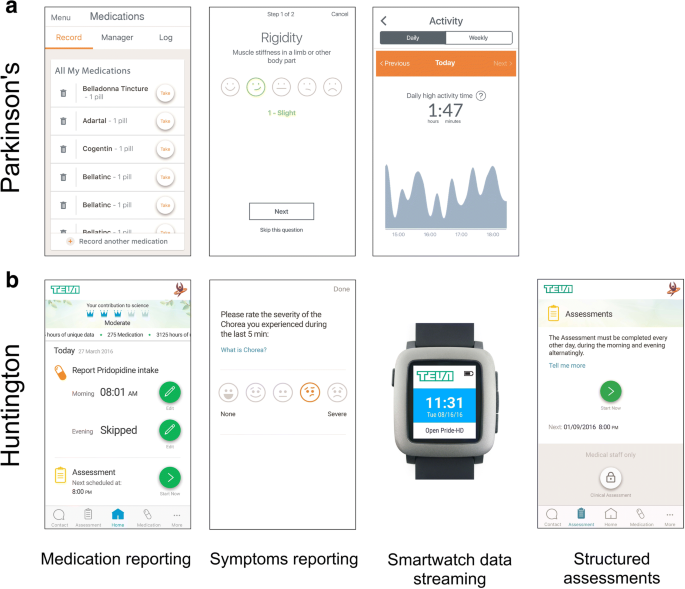
Characterizing patient compliance over six months in remote digital trials of Parkinson's and Huntington disease | BMC Medical Informatics and Decision Making | Full Text

PDF) Phase 1 dose-escalation study of the PARP inhibitor CEP-9722 as monotherapy or in combination with temozolomide in patients with solid tumors

Teva Branded Pharmaceutical Products R&D;, . An electronic inhaler add-on module which store and transmit information related to inhaler use (eModule) PDR0000401 FCC ID 2AJVSPDR0000401

Teva Branded Pharmaceutical Products R&D;, . An electronic inhaler add-on module which store and transmit information related to inhaler use (eModule) PDR0000401 FCC ID 2AJVSPDR0000401

Assessment of Alcohol-Induced Dose Dumping with a Hydrocodone Bitartrate Extended-Release Tablet Formulated with CIMA® Abuse Deterrence Technology – topic of research paper in Clinical medicine. Download scholarly article PDF and read for