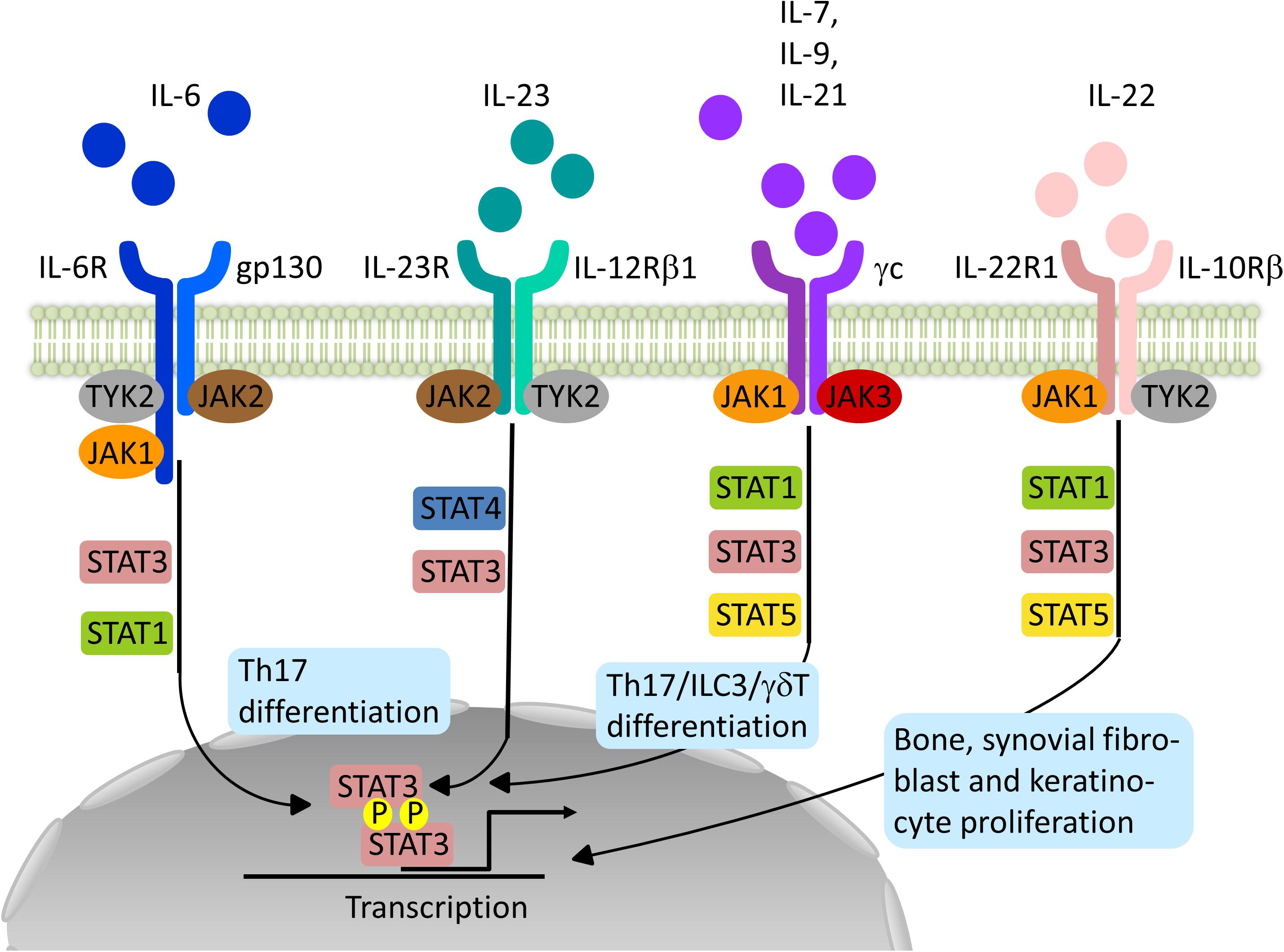
Frontiers | Impact of Janus Kinase Inhibition on the Treatment of Axial Spondyloarthropathies | Immunology
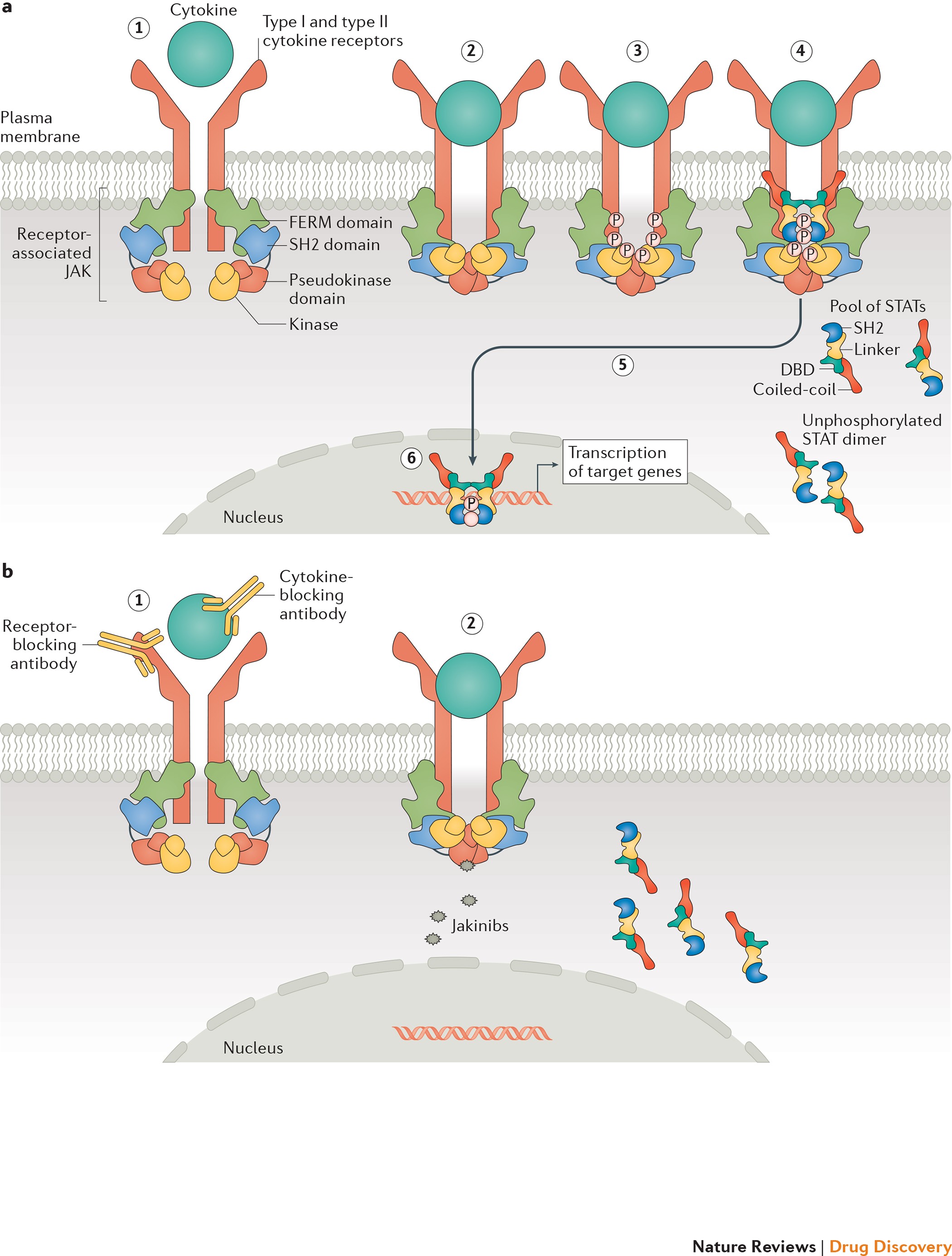
JAK inhibition as a therapeutic strategy for immune and inflammatory diseases | Nature Reviews Drug Discovery

Pfizer Announces FDA Approval of XELJANZ® XR (tofacitinib citrate) Extended-Release Tablets, the First and Only Once-Daily Oral JAK Inhibitor Treatment for Rheumatoid Arthritis | Business Wire

Janus kinase inhibitors in dermatology: Part II. A comprehensive review - Journal of the American Academy of Dermatology
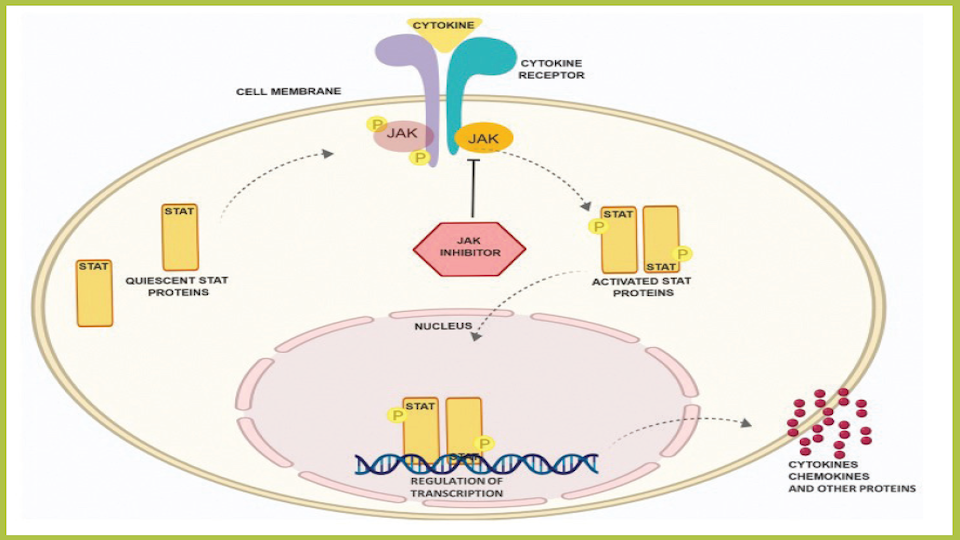
JAK Inhibitors Are Coming and They Are the Biggest Eczema Development in Years | National Eczema Association

Pharmas with up-and-coming JAK inhibitors face 'shrinking' potential after FDA crackdown | Fierce Biotech
Pfizer Xeljanz, Tofacitinib (tofacitinib citrate), JAK inhibitor, 11mg tablets, once daily treatment in sustained release tablet form, France Stock Photo - Alamy
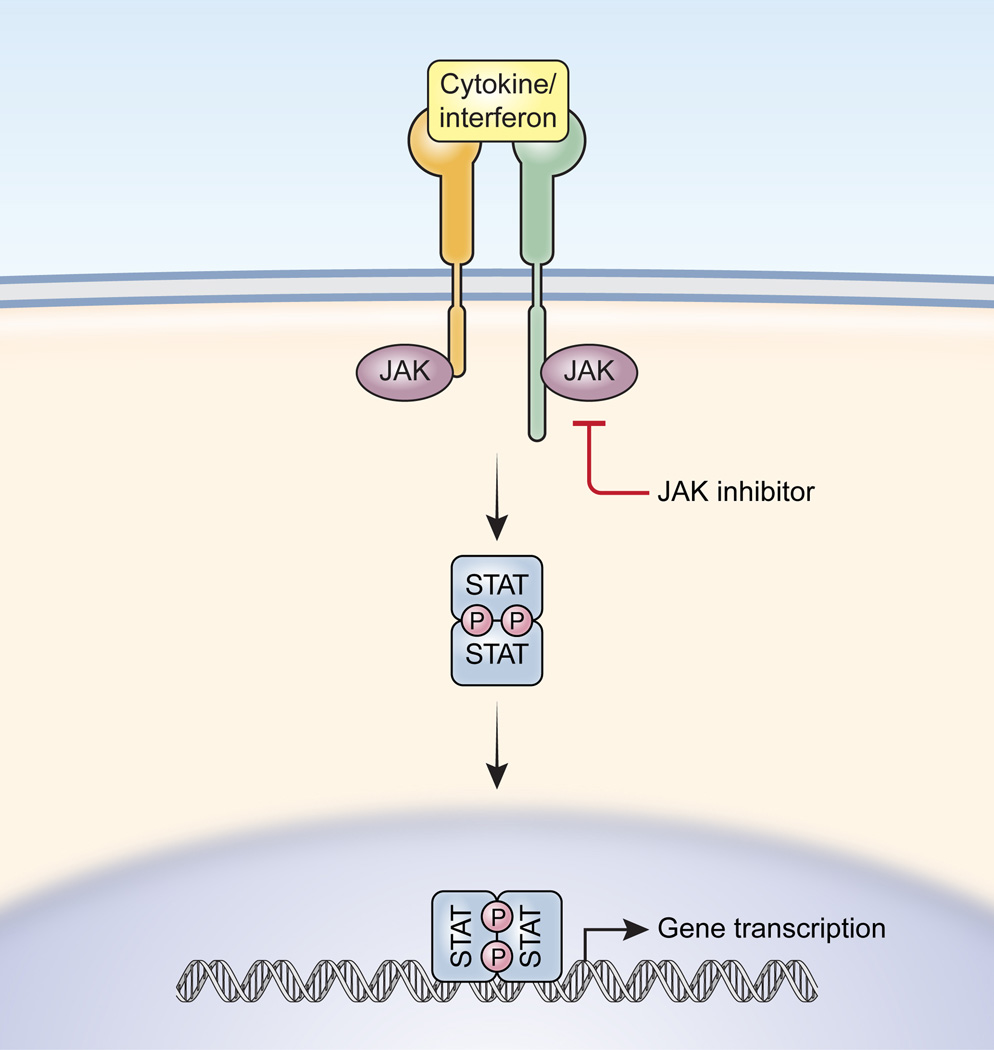







:max_bytes(150000):strip_icc()/jak-inhibitors-4706526-FINAL-3461d4fc6ce44f54801ef59489afa62c.png)

