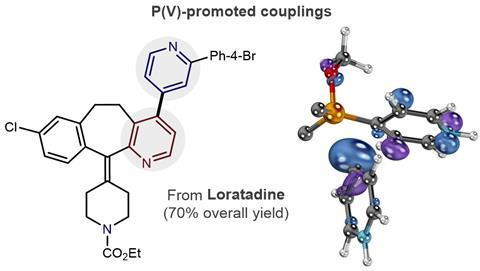
Palladium-catalyzed phosphorus–carbon bond formation: cross-coupling reactions of alkyl phosphinates with aryl, heteroaryl, alkenyl, benzylic, and allylic halides and triflates - ScienceDirect

Developing backbone-modified Mor-DalPhos ligand variants for use in palladium-catalyzed C–N and C–C cross-coupling
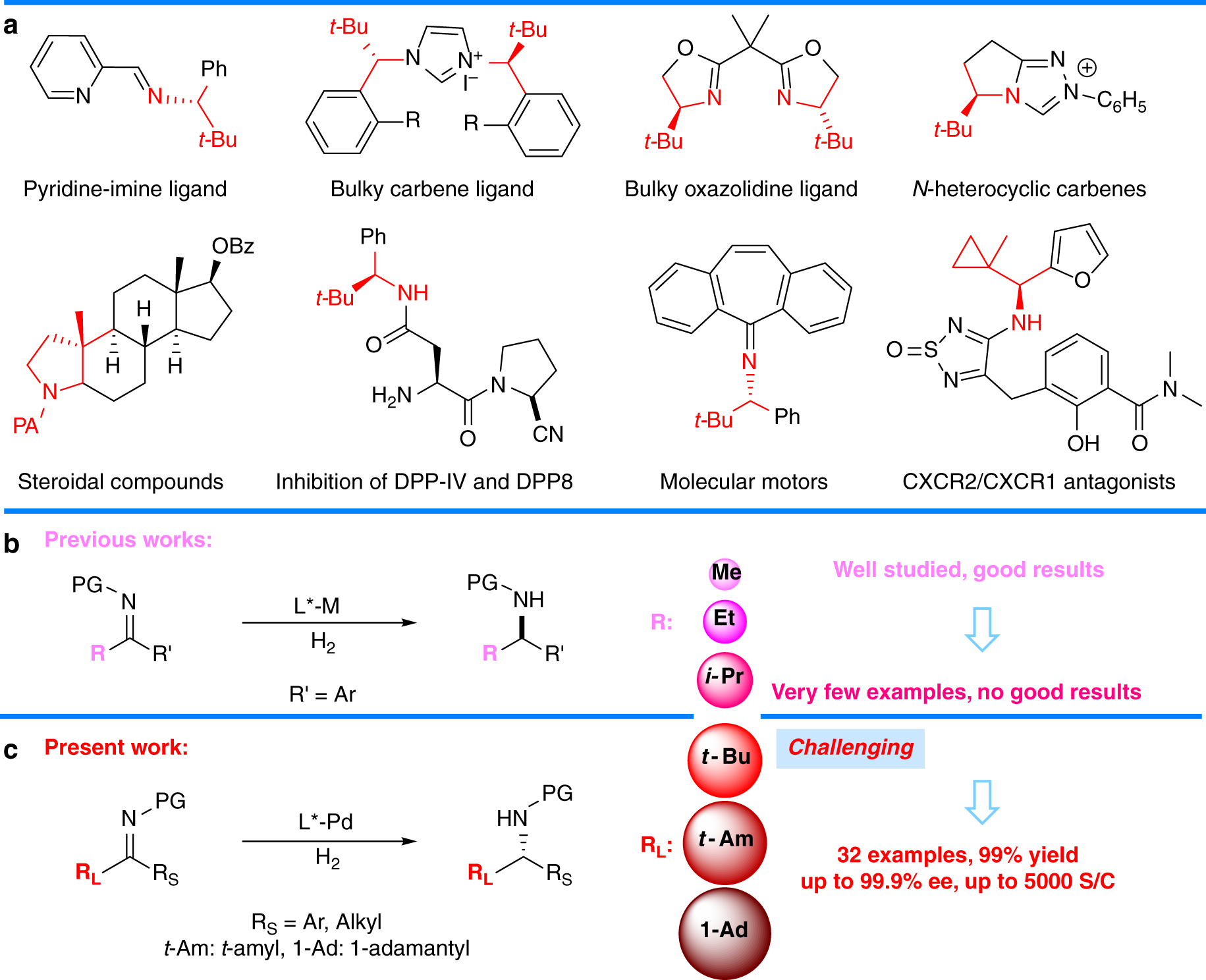
Pd(OAc)2-catalyzed asymmetric hydrogenation of sterically hindered N-tosylimines | Nature Communications

A Versatile Ligand Platform for Palladium‐ and Nickel‐Catalyzed Ethylene Copolymerization with Polar Monomers - Chen - 2018 - Angewandte Chemie International Edition - Wiley Online Library
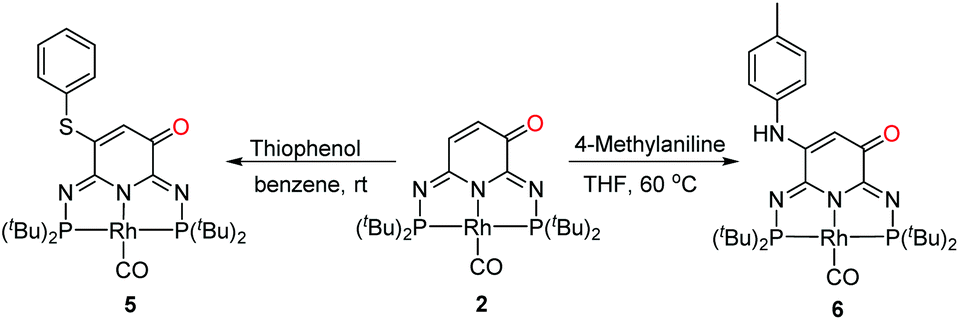
Ligand-centered reactivity of a pseudo-dearomatized phosphorus-nitrogen PN 3 P* rhodium complex towards molecular oxygen at room temperature - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QI01605H
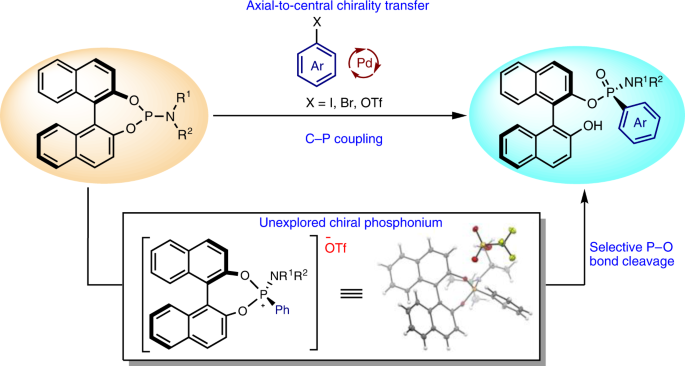
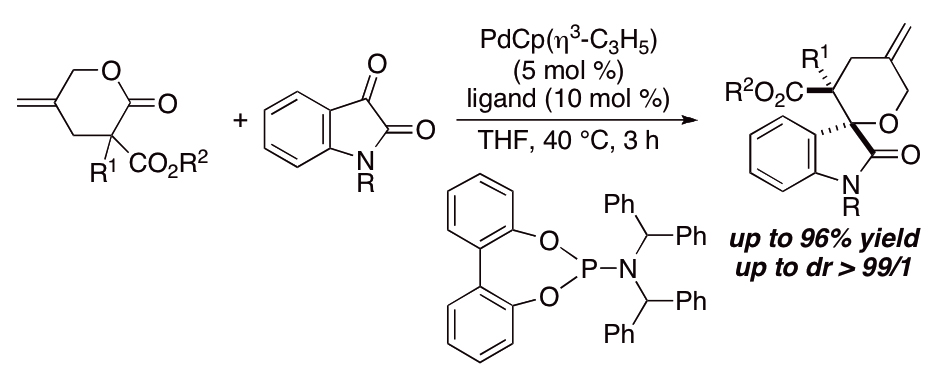
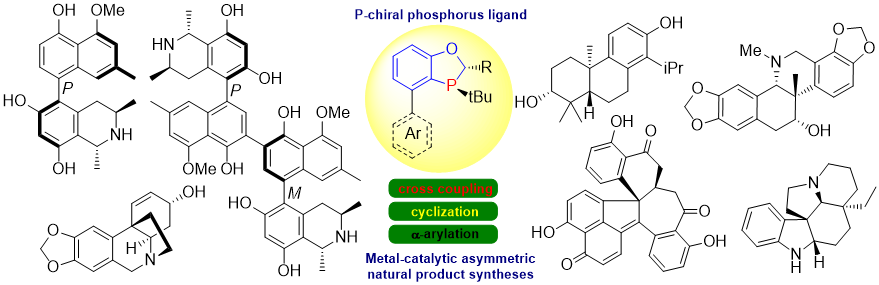
P-chirogenic phosphorus compounds by stereoselective Pd-catalysed arylation of phosphoramidites | Nature Catalysis

Developing Ligands for Palladium(II)-Catalyzed C–H Functionalization: Intimate Dialogue between Ligand and Substrate | The Journal of Organic Chemistry

Aerobic Heterogeneous Palladium-Catalyzed Oxidative Allenic C−H Arylation: Benzoquinone as a Direct Redox Mediator between O2 and Pd | CCS Chem

Synthesis, characterization, coordination chemistry, and luminescence studies of copper, silver, palladium, and platinum complexes with a phosphorus/nitrogen/phosphorus ligand - ScienceDirect

Agarose functionalized phosphorus ligand for stabilization of small-sized palladium and copper nanoparticles: efficient heterogeneous catalyst for Sonogashira reaction - ScienceDirect
Ligand-centered reactivity of a pseudo-dearomatized phosphorus-nitrogen PN 3 P* rhodium complex towards molecular oxygen at room temperature - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QI01605H
Palladium catalyzed cross-coupling reactions for phosphorus–carbon bond formation - Chemical Society Reviews (RSC Publishing)
![Phosphiranes as Ligands: Tungsten(0) and Palladium(0) Complexes of Phosphirano[1,2‐c][1,2,3]diazaphospholes - Maurer - 2010 - European Journal of Organic Chemistry - Wiley Online Library Phosphiranes as Ligands: Tungsten(0) and Palladium(0) Complexes of Phosphirano[1,2‐c][1,2,3]diazaphospholes - Maurer - 2010 - European Journal of Organic Chemistry - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/1efad1ce-971f-4d08-88ed-1b4a563eb5ea/mfig000.jpg)
Phosphiranes as Ligands: Tungsten(0) and Palladium(0) Complexes of Phosphirano[1,2‐c][1,2,3]diazaphospholes - Maurer - 2010 - European Journal of Organic Chemistry - Wiley Online Library

Monodentate phosphorus-coordinated palladium(II) complexes as new catalyst for Mizoroki-Heck reaction of aryl halides with electron-deficient olefins - ScienceDirect

Aerobic Heterogeneous Palladium-Catalyzed Oxidative Allenic C−H Arylation: Benzoquinone as a Direct Redox Mediator between O2 and Pd | CCS Chem
![Synthesis of 3-aryl-2-phosphinoimidazo[1,2- a ]pyridine ligands for use in palladium-catalyzed cross-coupling reactions - RSC Advances (RSC Publishing) DOI:10.1039/C9RA02200G Synthesis of 3-aryl-2-phosphinoimidazo[1,2- a ]pyridine ligands for use in palladium-catalyzed cross-coupling reactions - RSC Advances (RSC Publishing) DOI:10.1039/C9RA02200G](https://pubs.rsc.org/image/article/2019/RA/c9ra02200g/c9ra02200g-s1_hi-res.gif)
Synthesis of 3-aryl-2-phosphinoimidazo[1,2- a ]pyridine ligands for use in palladium-catalyzed cross-coupling reactions - RSC Advances (RSC Publishing) DOI:10.1039/C9RA02200G