Cropped hand of male scientist mixing chemical formula in test tube for experiment at laboratory — glove, scientific Sample - Stock Photo | #474276040

Research experiment, Scientist mixing a chemical formula in a test tube — examination, laboratory - Stock Photo | #268292884
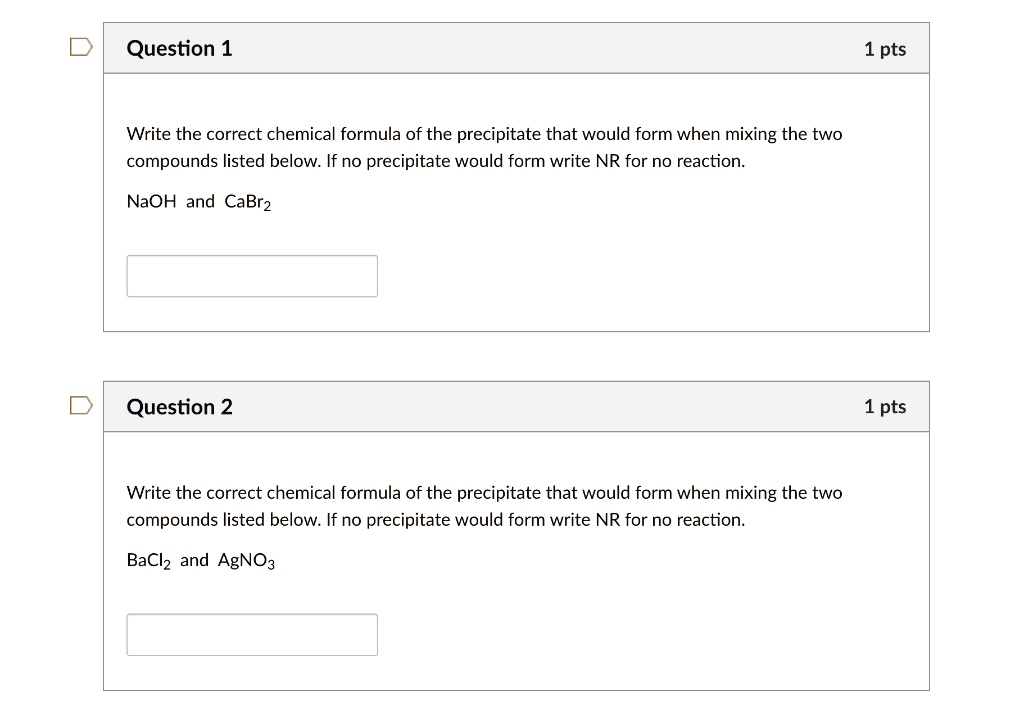
SOLVED:Question 1 1 pts Write the correct chemical formula of the precipitate that would form when mixing the two compounds listed below: If no precipitate would form write NR for no reaction:

A ehite chemical compound becomes hard on mixing proper quantity of water. It is also used to maintain joints

Acids and Bases Calculating Excess. Mixing strong acids and bases During an experiment, a student pours 25.0 mL of 1.40 mol/L nitric acid into a beaker. - ppt download

Biotechnology Research, female scientist mixing a chemical formula, Stock Photo, Picture And Royalty Free Image. Pic. WES-ABRF00167 | agefotostock
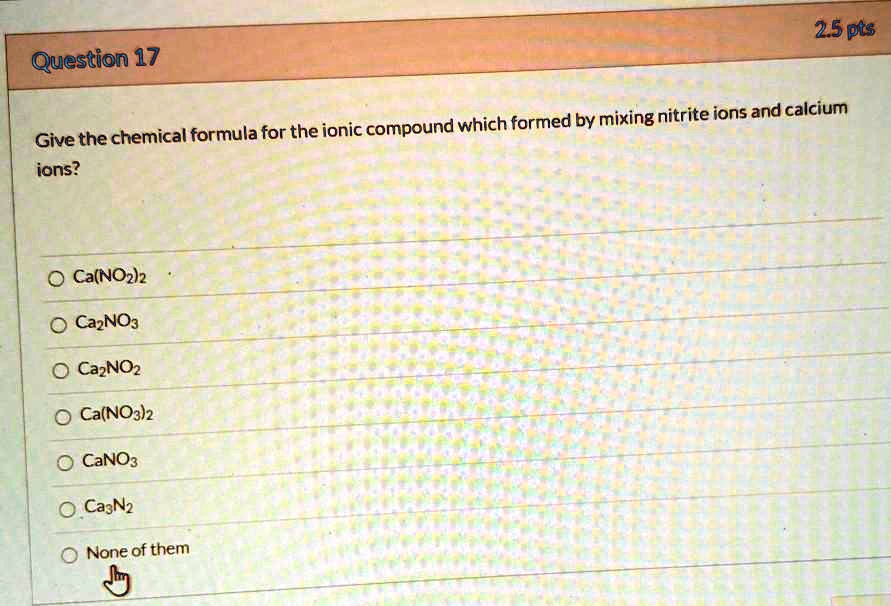





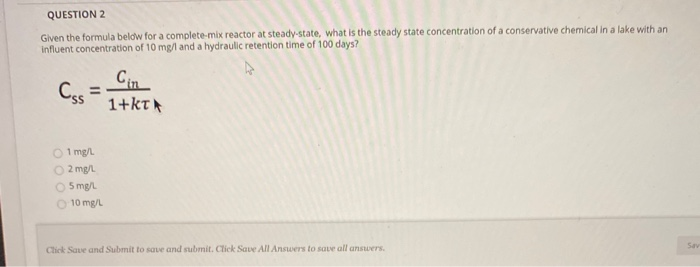




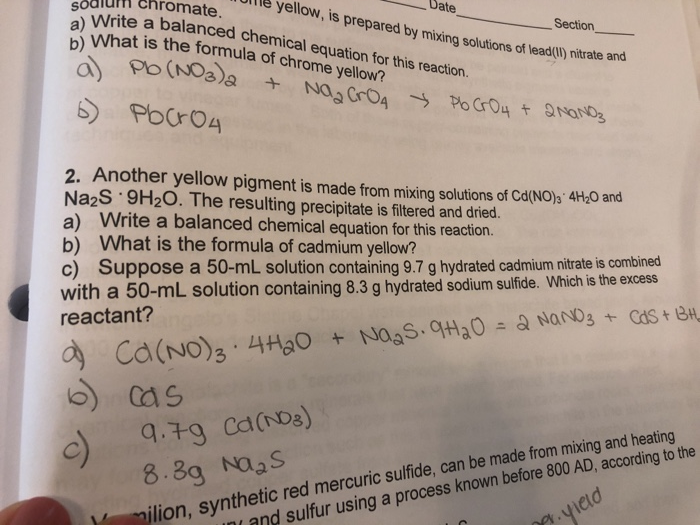



.jpg)

/173637959-58befc655f9b58af5c9acab8.jpg)